
Q = the heat exchanged between a system and its surroundings (outside the system) U = the total change in internal energy within the system The Change in Internal Energy Formula is: The potential energy remains stored in the form of chemical bonds, attraction, or repulsion.Īlso, kinetic energy, which is because of the motion, translation, rotation, and vibration of particles within the system. The internal energy also involves the potential energy. The total internal energy formula is the sum of transnational energies, rotational energies, binding energies, and chemical energies of all the particles in the system. 
The equation (2) is also called the total internal energy formula or the change in internal energy equation) Total Internal Energy Formula Rearranging the equation (1) to get the formula for change in internal energy: On this page, we will understand the internal energy formula, internal energy formula, ideal gas, specific internal energy formula, change in internal energy equation, and the total internal energy formula.Īs per the first law of thermodynamics, the energy of the universe is invariant.Īlso, the change in the internal energy of a system equals the total of the heat transferred and the work done.īesides this, the heat added/flown is equal to the sum of the change in the internal energy of the system and the PV work done. The change in internal energy equals the difference in the heat flow in a system and the work done by/on the system (PV) and the change in internal energy formula helps us calculate the same. We can calculate the same by the internal energy formula. In short, internal energy is the energy associated with the random motion or disordered arrangement of particles within the system, which is measured in KJ or Joule. It is the energy of a substance due to kinetic and potential energies that are associated with the random motion of all the particles that make up the substance. 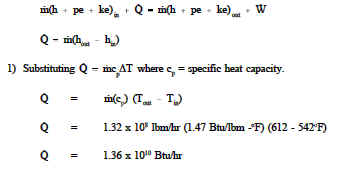
Internal energy is also called thermal energy. It is symbolized by the English letter ‘U.’ The energy contained within the system is called internal energy.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
